CHEMISTRY BEHIND THE REACTION
The Diet Coke and Mentos experiment may be one of the most popular experiments, but of the people who perform this experiment not that many actually understand the chemistry concepts behind it. There have been several disputed theories as to why Diet Coke erupts in a geyser when introduced to Mentos, but the most prominent and most supported are that of nucleation and a chemical reaction between the soda and Mentos. Many notable sources including Scientific American, American Chemical Society, and Discover Chanel's Mythbusters have created content in regards to this reaction with varying beliefs and assumptions.
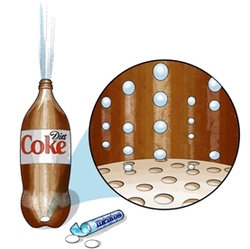
The general premise of this reaction is based upon nucleation - nucleation being the process in which particles in the gaseous state group in a pore or rough surface forming a large bubble of gas. In the case of this experiment, C02 particles from the Diet Coke are trapped in the rough pitted texture of the Mentos forming larger bubbles of C02 gas, which float to the surface pushing soda up and shooting it out the mouth of the bottle in an eruption fashion creating a geyser or fountain of soda.
The general premise of this reaction is based upon nucleation - nucleation being the process in which particles in the gaseous state group in a pore or rough surface forming a large bubble of gas. In the case of this experiment, C02 particles from the Diet Coke are trapped in the rough pitted texture of the Mentos forming larger bubbles of C02 gas, which float to the surface pushing soda up and shooting it out the mouth of the bottle in an eruption fashion creating a geyser or fountain of soda.
|
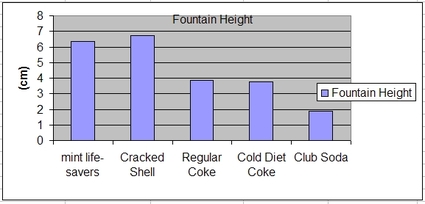
The theory that nucleation causes this reaction is well defended as a variety of different variables have been tested proving so that it truly is the nucleation resulting in the geyser. The Mythbusters altered the variables of the type of soda and the texture of the Mentos on separate occasions through comparing the original reaction to reactions with soda water and smooth Mentos to find that the reaction is significantly lesser than original. As the soda water has less carbonation, less C02 particles are able to be trapped in nucleation meaning that the reaction should be less according to nucleation, which was proven correct. In addition, the variable of texture of the Mentos was proved according to the fact that when smooth Mentos were used instead, little to no reaction occurred proving that nucleation is present in this reaction. The following graph represents the results changed upon the alteration of particular variables previously mentioned.
In addition to the nucleation that occurs, a chemical reaction between chemicals from the Mentos and Diet Coke react to result in a larger fountain, explaining why Diet Coke is more effective than tonic water or non-Diet Coke. The ingredients Gum Arabic and Gelatin found in the Mentos react with the Benzoate as well as the Aspritane in the Diet Coke, the reaction lowers the surface tension of the cola at the mouth of the bottle. The reduction of the surface tension will cause for a larger fountain of soda to erupt, as the departing molecules use less energy to break the surface tension meaning they will go higher in the air. This explains why diet soda works better than non-diet soda as the Aspritane reacts better with the Mentos than sugar causing the surface tension to be further reduced than normal soda meaning that the fountain will go significantly higher. It is the combination of these two concepts that result in the fountain that is especially large between Diet Coke and Mentos. The chemical reaction between the Mentos and Diet Coke reduce the surface tension on the top of the water allowing the gas created through nucleation to waste less energy breaking the surface tension as it dispels the soda out the mouth of the bottle in what is observed as the fountain or geyser. It may be said that these two concepts "work together" to result in the over 1-meter fountain of soda in this nostalgia inspiring experiment. |
|